1
JEE Main 2019 (Online) 9th January Evening Slot
MCQ (Single Correct Answer)
+4
-1

A 15 g mass of nitrogen gas is enclosed in a vessel at a temperature 27oC. Amount of heat transferred to the gas, so that rms velocity of molecules is doubled, is about : [Take R = 8.3 J/K mole]
2
JEE Main 2019 (Online) 9th January Morning Slot
MCQ (Single Correct Answer)
+4
-1

Temperature difference of 120oC is maintained between ends of a uniform rod AB of length 2L. Another bent rod PQ, of same cross-section as AB and length $${{3L} \over 2},$$ is connected across AB (see figure). In steady state, temperature difference between P and Q will be close to :


3
JEE Main 2019 (Online) 9th January Morning Slot
MCQ (Single Correct Answer)
+4
-1

A mixture of 2 moles of helium gas (atomic mass = 4 u), and 1 mole of argon gas (atomic mass = 40 u) is kept at 300 K in a container. The ratio of their rms speeds $$\left[ {{{{V_{rms}}\,(helium)} \over {{V_{rms}}\,(\arg on)}}} \right],$$ is close to :
4
JEE Main 2019 (Online) 9th January Morning Slot
MCQ (Single Correct Answer)
+4
-1

A gas can be taken from A to B via two different processes ACB and ADB.
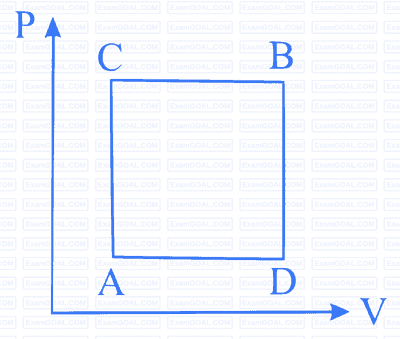
When path ACB is used 60 J of heat flows into the system and 30 J of work is done by the system. If path ADB is used work done by the system is 10 J. The heat Flow into the system in path ADB is :

When path ACB is used 60 J of heat flows into the system and 30 J of work is done by the system. If path ADB is used work done by the system is 10 J. The heat Flow into the system in path ADB is :
JEE Main Subjects
Browse all chapters by subject
Physics
Mechanics
Units & Measurements Vector Algebra Motion in a Straight Line Motion in a Plane Circular Motion Laws of Motion Work Power & Energy Center of Mass and Collision Rotational Motion Properties of Matter Heat and Thermodynamics Simple Harmonic Motion Waves Gravitation
Electricity
Electrostatics Current Electricity Capacitor Magnetic Effect of Current Magnetic Properties of Matter Electromagnetic Induction Alternating Current Electromagnetic Waves
Optics
Modern Physics
Chemistry
Physical Chemistry
Some Basic Concepts of Chemistry Structure of Atom Redox Reactions Gaseous State Chemical Equilibrium Ionic Equilibrium Solutions Thermodynamics Electrochemistry Chemical Kinetics and Nuclear Chemistry Solid State Surface Chemistry
Inorganic Chemistry
Periodic Table & Periodicity Chemical Bonding & Molecular Structure Isolation of Elements Hydrogen s-Block Elements p-Block Elements d and f Block Elements Coordination Compounds Salt Analysis Environmental Chemistry
Organic Chemistry
Mathematics
Algebra
Sets and Relations Logarithm Quadratic Equation and Inequalities Sequences and Series Mathematical Induction Binomial Theorem Matrices and Determinants Permutations and Combinations Probability Vector Algebra 3D Geometry Complex Numbers Statistics Mathematical Reasoning
Trigonometry
Trigonometric Ratio and Identites Trigonometric Equations Inverse Trigonometric Functions Properties of Triangle Height and Distance
Coordinate Geometry
Calculus