1
JEE Main 2020 (Online) 5th September Evening Slot
MCQ (Single Correct Answer)
+4
-1

Two different wires having lengths L1 and L2,
and respective temperature coefficient of linear
expansion $$\alpha $$1 and $$\alpha $$2, are joined end-to-end.
Then the effective temperature coefficient of
linear expansion is :
2
JEE Main 2020 (Online) 5th September Evening Slot
MCQ (Single Correct Answer)
+4
-1

In an adiabatic process, the density of a
diatomic gas becomes 32 times its initial value.
The final pressure of the gas is found to be n
times the initial pressure. The value of n is :
3
JEE Main 2020 (Online) 5th September Morning Slot
MCQ (Single Correct Answer)
+4
-1

A bullet of mass 5 g, travelling with a speed of
210 m/s, strikes a fixed wooden target. One half
of its kinetic energy is converted into heat in
the bullet while the other half is converted into
heat in the wood. The rise of temperature of
the bullet if the specific heat of its material is
0.030 cal/(g – oC) (1 cal = 4.2 × 107 ergs) close
to :
4
JEE Main 2020 (Online) 5th September Morning Slot
MCQ (Single Correct Answer)
+4
-1

Three different processes that can occur in an
ideal monoatomic gas are shown in the P vs V
diagram. The paths are labelled as A $$ \to $$ B,
A $$ \to $$ C and A $$ \to $$ D. The change in internal
energies during these process are taken as
EAB, EAC and EAD and the work done as WAB,
WAC and WAD.
The correct relation between these parameters are :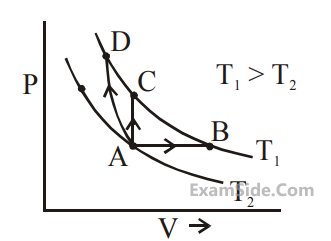
The correct relation between these parameters are :

JEE Main Subjects
Browse all chapters by subject
Physics
Mechanics
Units & Measurements Vector Algebra Motion in a Straight Line Motion in a Plane Circular Motion Laws of Motion Work Power & Energy Center of Mass and Collision Rotational Motion Properties of Matter Heat and Thermodynamics Simple Harmonic Motion Waves Gravitation
Electricity
Electrostatics Current Electricity Capacitor Magnetic Effect of Current Magnetic Properties of Matter Electromagnetic Induction Alternating Current Electromagnetic Waves
Optics
Modern Physics
Chemistry
Physical Chemistry
Some Basic Concepts of Chemistry Structure of Atom Redox Reactions Gaseous State Chemical Equilibrium Ionic Equilibrium Solutions Thermodynamics Electrochemistry Chemical Kinetics and Nuclear Chemistry Solid State Surface Chemistry
Inorganic Chemistry
Periodic Table & Periodicity Chemical Bonding & Molecular Structure Isolation of Elements Hydrogen s-Block Elements p-Block Elements d and f Block Elements Coordination Compounds Salt Analysis Environmental Chemistry
Organic Chemistry
Mathematics
Algebra
Sets and Relations Logarithm Quadratic Equation and Inequalities Sequences and Series Mathematical Induction Binomial Theorem Matrices and Determinants Permutations and Combinations Probability Vector Algebra 3D Geometry Complex Numbers Statistics Mathematical Reasoning
Trigonometry
Trigonometric Ratio and Identites Trigonometric Equations Inverse Trigonometric Functions Properties of Triangle Height and Distance
Coordinate Geometry
Calculus