1
JEE Main 2013 (Offline)
MCQ (Single Correct Answer)
+4
-1
Assume that a drop of liquid evaporates by decreases in its surface energy, so that its temperature remains unchanged. What should be the minimum radius of the drop for this to be possible ? The surface tension is $$T,$$ density of liquid is $$\rho $$ and $$L$$ is its latent heat of vaporization.
2
JEE Main 2013 (Offline)
MCQ (Single Correct Answer)
+4
-1
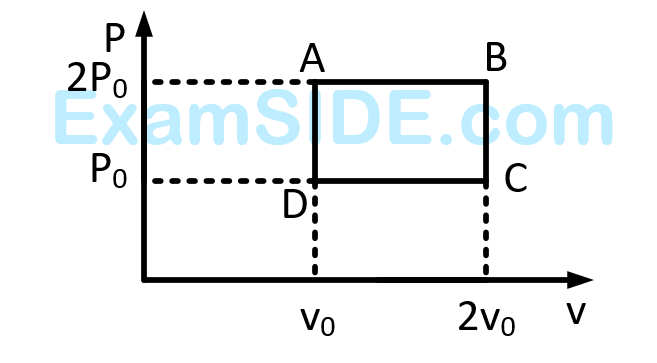
The above $$p$$-$$v$$ diagram represents the thermodynamic cycle of an engine, operating with an ideal monatomic gas. The amount of heat, extracted from the source in a single cycle is
3
AIEEE 2012
MCQ (Single Correct Answer)
+4
-1
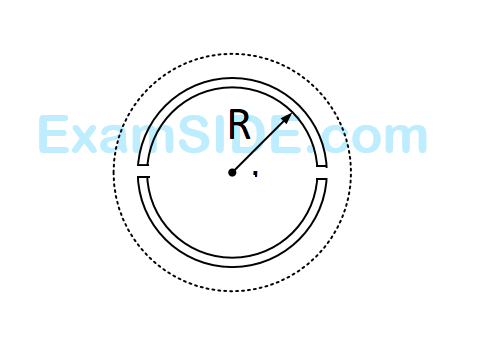
A wooden wheel of radius $$R$$ is made of two semicircular part (see figure). The two parts are held together by a ring made of a metal strip of cross sectional area $$S$$ and length $$L.$$ $$L$$ is slightly less than $$2\pi R.$$ To fit the ring on the wheel, it is heated so that its temperature rises by $$\Delta T$$ and it just steps over the wheel. As it cools down to surrounding temperature, it process the semicircular parts together. If the coefficient of linear expansion of the metal is $$\alpha $$, and its Young's modulus is $$Y,$$ the force that one part of the wheel applies on the other part is :

4
AIEEE 2012
MCQ (Single Correct Answer)
+4
-1
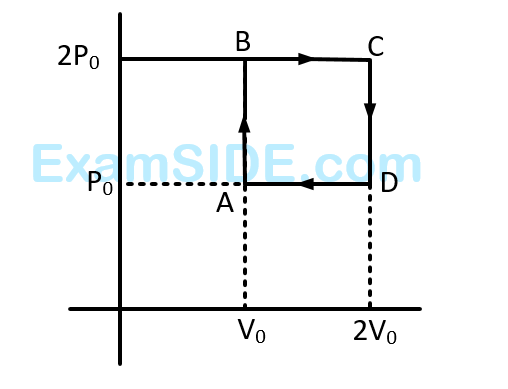
Helium gas goes through a cycle $$ABCD$$ (consisting of two isochoric and isobaric lines) as shown in figure efficiency of this cycle is nearly : (Assume the gas to be close to ideal gas)

JEE Main Subjects
Browse all chapters by subject
Physics
Mechanics
Units & Measurements Vector Algebra Motion in a Straight Line Motion in a Plane Circular Motion Laws of Motion Work Power & Energy Center of Mass and Collision Rotational Motion Properties of Matter Heat and Thermodynamics Simple Harmonic Motion Waves Gravitation
Electricity
Electrostatics Current Electricity Capacitor Magnetic Effect of Current Magnetic Properties of Matter Electromagnetic Induction Alternating Current Electromagnetic Waves
Optics
Modern Physics
Chemistry
Physical Chemistry
Some Basic Concepts of Chemistry Structure of Atom Redox Reactions Gaseous State Chemical Equilibrium Ionic Equilibrium Solutions Thermodynamics Electrochemistry Chemical Kinetics and Nuclear Chemistry Solid State Surface Chemistry
Inorganic Chemistry
Periodic Table & Periodicity Chemical Bonding & Molecular Structure Isolation of Elements Hydrogen s-Block Elements p-Block Elements d and f Block Elements Coordination Compounds Salt Analysis Environmental Chemistry
Organic Chemistry
Mathematics
Algebra
Sets and Relations Logarithm Quadratic Equation and Inequalities Sequences and Series Mathematical Induction Binomial Theorem Matrices and Determinants Permutations and Combinations Probability Vector Algebra 3D Geometry Complex Numbers Statistics Mathematical Reasoning
Trigonometry
Trigonometric Ratio and Identites Trigonometric Equations Inverse Trigonometric Functions Properties of Triangle Height and Distance
Coordinate Geometry
Calculus