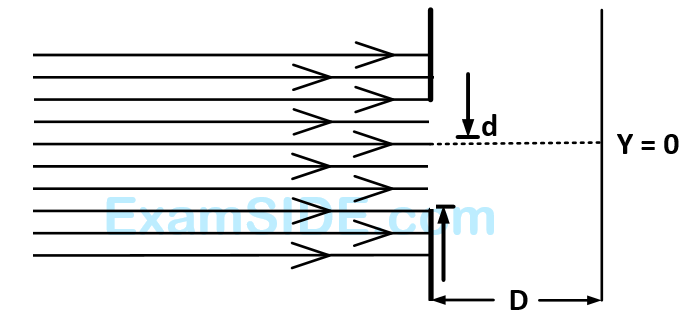
Which of the following graphs can be expected to represent the number of electrons $$'N'$$ detected as a function of the detector position $$'y'\left( {y = 0} \right.$$ corresponds to the middle of the slit$$\left. \, \right)$$
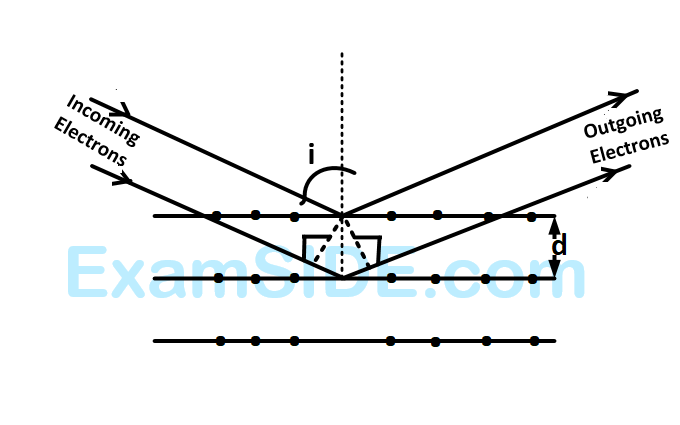
If a strong diffraction peak is observed when electrons are incident at an angle $$'i'$$ from the normal to the crystal planes with distance $$'d'$$ between them (see figure), de Broglie wavelength $${\lambda _{dB}}$$ of electrons can be calculated by the relationship ($$n$$ is an integer)

Electrons accelerated by potential $$V$$ are diffracted from a crystal. If $$d = 1\mathop A\limits^ \circ $$ and $$i = {30^ \circ },\,\,\,V$$ should be about
$$\left( {h = 6.6 \times {{10}^{ - 34}}Js,{m_e} = 9.1 \times {{10}^{ - 31}}kg,\,e = 1.6 \times {{10}^{ - 19}}C} \right)$$
JEE Main Subjects
Browse all chapters by subject



