1
JEE Main 2019 (Online) 11th January Evening Slot
MCQ (Single Correct Answer)
+4
-1

25 ml of the given HCl solution requires 30 mL of 0.1 M sodium carbonate solution. What is the volume of this HCl solution required to titrate 30 mL of 0.2 M aqueous NaOH solutions ?
2
JEE Main 2019 (Online) 11th January Evening Slot
MCQ (Single Correct Answer)
+4
-1

The major product obtained in the following conversion is
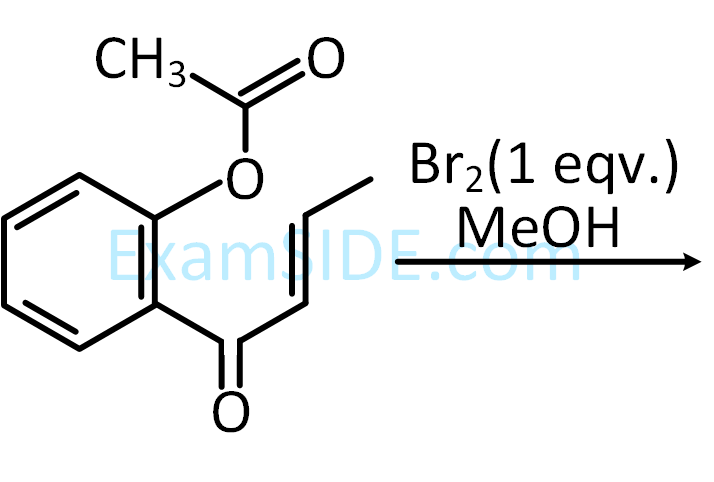

3
JEE Main 2019 (Online) 11th January Evening Slot
MCQ (Single Correct Answer)
+4
-1

Let S = {1, 2, . . . . . ., 20}. A subset B of S is said to be "nice", if the sum of the elements of B is 203. Then the probability that a randonly chosen subset of S is "nice" is :
4
JEE Main 2019 (Online) 11th January Evening Slot
MCQ (Single Correct Answer)
+4
-1

Let K be the set of all real values of x where the function f(x) = sin |x| – |x| + 2(x – $$\pi $$) cos |x| is not differentiable. Then the set K is equal to :
Paper Analysis
Total Questions
Chemistry 20
Mathematics 22
Physics 28
More Papers of JEE Main
JEE Main 2026 (Online) 8th April Evening Shift JEE Main 2026 (Online) 6th April Evening Shift JEE Main 2026 (Online) 6th April Morning Shift JEE Main 2026 (Online) 5th April Evening Shift JEE Main 2026 (Online) 5th April Morning Shift JEE Main 2026 (Online) 4th April Evening Shift JEE Main 2026 (Online) 4th April Morning Shift JEE Main 2026 (Online) 2nd April Evening Shift JEE Main 2026 (Online) 2nd April Morning Shift JEE Main 2026 (Online) 28th January Evening Shift JEE Main 2026 (Online) 28th January Morning Shift JEE Main 2026 (Online) 24th January Evening Shift JEE Main 2026 (Online) 24th January Morning Shift JEE Main 2026 (Online) 23rd January Evening Shift JEE Main 2026 (Online) 23rd January Morning Shift JEE Main 2026 (Online) 22nd January Evening Shift JEE Main 2026 (Online) 22nd January Morning Shift JEE Main 2026 (Online) 21st January Evening Shift JEE Main 2026 (Online) 21st January Morning Shift JEE Main 2025 (Online) 8th April Evening Shift JEE Main 2025 (Online) 7th April Evening Shift JEE Main 2025 (Online) 7th April Morning Shift JEE Main 2025 (Online) 4th April Evening Shift JEE Main 2025 (Online) 4th April Morning Shift JEE Main 2025 (Online) 3rd April Evening Shift JEE Main 2025 (Online) 3rd April Morning Shift JEE Main 2025 (Online) 2nd April Evening Shift JEE Main 2025 (Online) 2nd April Morning Shift JEE Main 2025 (Online) 29th January Evening Shift JEE Main 2025 (Online) 29th January Morning Shift JEE Main 2025 (Online) 28th January Evening Shift JEE Main 2025 (Online) 28th January Morning Shift JEE Main 2025 (Online) 24th January Evening Shift JEE Main 2025 (Online) 24th January Morning Shift JEE Main 2025 (Online) 23rd January Evening Shift JEE Main 2025 (Online) 23rd January Morning Shift JEE Main 2025 (Online) 22nd January Evening Shift JEE Main 2025 (Online) 22nd January Morning Shift JEE Main 2024 (Online) 9th April Evening Shift JEE Main 2024 (Online) 9th April Morning Shift JEE Main 2024 (Online) 8th April Evening Shift JEE Main 2024 (Online) 8th April Morning Shift JEE Main 2024 (Online) 6th April Evening Shift JEE Main 2024 (Online) 6th April Morning Shift JEE Main 2024 (Online) 5th April Evening Shift JEE Main 2024 (Online) 5th April Morning Shift JEE Main 2024 (Online) 4th April Evening Shift JEE Main 2024 (Online) 4th April Morning Shift JEE Main 2024 (Online) 1st February Evening Shift JEE Main 2024 (Online) 1st February Morning Shift JEE Main 2024 (Online) 31st January Evening Shift JEE Main 2024 (Online) 31st January Morning Shift JEE Main 2024 (Online) 30th January Evening Shift JEE Main 2024 (Online) 30th January Morning Shift JEE Main 2024 (Online) 29th January Evening Shift JEE Main 2024 (Online) 29th January Morning Shift JEE Main 2024 (Online) 27th January Evening Shift JEE Main 2024 (Online) 27th January Morning Shift JEE Main 2023 (Online) 15th April Morning Shift JEE Main 2023 (Online) 13th April Evening Shift JEE Main 2023 (Online) 13th April Morning Shift JEE Main 2023 (Online) 12th April Morning Shift JEE Main 2023 (Online) 11th April Evening Shift JEE Main 2023 (Online) 11th April Morning Shift JEE Main 2023 (Online) 10th April Evening Shift JEE Main 2023 (Online) 10th April Morning Shift JEE Main 2023 (Online) 8th April Evening Shift JEE Main 2023 (Online) 8th April Morning Shift JEE Main 2023 (Online) 6th April Evening Shift JEE Main 2023 (Online) 6th April Morning Shift JEE Main 2023 (Online) 1st February Evening Shift JEE Main 2023 (Online) 1st February Morning Shift JEE Main 2023 (Online) 31st January Evening Shift JEE Main 2023 (Online) 31st January Morning Shift JEE Main 2023 (Online) 30th January Evening Shift JEE Main 2023 (Online) 30th January Morning Shift JEE Main 2023 (Online) 29th January Evening Shift JEE Main 2023 (Online) 29th January Morning Shift JEE Main 2023 (Online) 25th January Evening Shift JEE Main 2023 (Online) 25th January Morning Shift JEE Main 2023 (Online) 24th January Evening Shift JEE Main 2023 (Online) 24th January Morning Shift JEE Main 2022 (Online) 29th July Evening Shift JEE Main 2022 (Online) 29th July Morning Shift JEE Main 2022 (Online) 28th July Evening Shift JEE Main 2022 (Online) 28th July Morning Shift JEE Main 2022 (Online) 27th July Evening Shift JEE Main 2022 (Online) 27th July Morning Shift JEE Main 2022 (Online) 26th July Evening Shift JEE Main 2022 (Online) 26th July Morning Shift JEE Main 2022 (Online) 25th July Evening Shift JEE Main 2022 (Online) 25th July Morning Shift JEE Main 2022 (Online) 30th June Morning Shift JEE Main 2022 (Online) 29th June Evening Shift JEE Main 2022 (Online) 29th June Morning Shift JEE Main 2022 (Online) 28th June Evening Shift JEE Main 2022 (Online) 28th June Morning Shift JEE Main 2022 (Online) 27th June Evening Shift JEE Main 2022 (Online) 27th June Morning Shift JEE Main 2022 (Online) 26th June Evening Shift JEE Main 2022 (Online) 26th June Morning Shift JEE Main 2022 (Online) 25th June Evening Shift JEE Main 2022 (Online) 25th June Morning Shift JEE Main 2022 (Online) 24th June Evening Shift JEE Main 2022 (Online) 24th June Morning Shift JEE Main 2021 (Online) 1st September Evening Shift JEE Main 2021 (Online) 31st August Evening Shift JEE Main 2021 (Online) 31st August Morning Shift JEE Main 2021 (Online) 27th August Evening Shift JEE Main 2021 (Online) 27th August Morning Shift JEE Main 2021 (Online) 26th August Evening Shift JEE Main 2021 (Online) 26th August Morning Shift JEE Main 2021 (Online) 27th July Evening Shift JEE Main 2021 (Online) 27th July Morning Shift JEE Main 2021 (Online) 25th July Evening Shift JEE Main 2021 (Online) 25th July Morning Shift JEE Main 2021 (Online) 22th July Evening Shift JEE Main 2021 (Online) 20th July Evening Shift JEE Main 2021 (Online) 20th July Morning Shift JEE Main 2021 (Online) 18th March Evening Shift JEE Main 2021 (Online) 18th March Morning Shift JEE Main 2021 (Online) 17th March Evening Shift JEE Main 2021 (Online) 17th March Morning Shift JEE Main 2021 (Online) 16th March Evening Shift JEE Main 2021 (Online) 16th March Morning Shift JEE Main 2021 (Online) 26th February Evening Shift JEE Main 2021 (Online) 26th February Morning Shift JEE Main 2021 (Online) 25th February Evening Shift JEE Main 2021 (Online) 25th February Morning Shift JEE Main 2021 (Online) 24th February Evening Shift JEE Main 2021 (Online) 24th February Morning Shift JEE Main 2020 (Online) 6th September Evening Slot JEE Main 2020 (Online) 6th September Morning Slot JEE Main 2020 (Online) 5th September Evening Slot JEE Main 2020 (Online) 5th September Morning Slot JEE Main 2020 (Online) 4th September Evening Slot JEE Main 2020 (Online) 4th September Morning Slot JEE Main 2020 (Online) 3rd September Evening Slot JEE Main 2020 (Online) 3rd September Morning Slot JEE Main 2020 (Online) 2nd September Evening Slot JEE Main 2020 (Online) 2nd September Morning Slot JEE Main 2020 (Online) 9th January Evening Slot JEE Main 2020 (Online) 9th January Morning Slot JEE Main 2020 (Online) 8th January Evening Slot JEE Main 2020 (Online) 8th January Morning Slot JEE Main 2020 (Online) 7th January Evening Slot JEE Main 2020 (Online) 7th January Morning Slot JEE Main 2019 (Online) 12th April Evening Slot JEE Main 2019 (Online) 12th April Morning Slot JEE Main 2019 (Online) 10th April Evening Slot JEE Main 2019 (Online) 10th April Morning Slot JEE Main 2019 (Online) 9th April Evening Slot JEE Main 2019 (Online) 9th April Morning Slot JEE Main 2019 (Online) 8th April Evening Slot JEE Main 2019 (Online) 8th April Morning Slot JEE Main 2019 (Online) 12th January Evening Slot JEE Main 2019 (Online) 12th January Morning Slot JEE Main 2019 (Online) 11th January Evening Slot JEE Main 2019 (Online) 11th January Morning Slot JEE Main 2019 (Online) 10th January Evening Slot JEE Main 2019 (Online) 10th January Morning Slot JEE Main 2019 (Online) 9th January Evening Slot JEE Main 2019 (Online) 9th January Morning Slot JEE Main 2018 (Online) 16th April Morning Slot JEE Main 2018 (Offline) JEE Main 2018 (Online) 15th April Evening Slot JEE Main 2018 (Online) 15th April Morning Slot JEE Main 2017 (Online) 9th April Morning Slot JEE Main 2017 (Online) 8th April Morning Slot JEE Main 2017 (Offline) JEE Main 2016 (Online) 10th April Morning Slot JEE Main 2016 (Online) 9th April Morning Slot JEE Main 2016 (Offline) JEE Main 2015 (Offline) JEE Main 2014 (Offline) JEE Main 2013 (Offline) AIEEE 2012 AIEEE 2011 AIEEE 2010 AIEEE 2009 AIEEE 2008 AIEEE 2007 AIEEE 2006 AIEEE 2005 AIEEE 2004 AIEEE 2003 AIEEE 2002
JEE Main Papers
All year-wise previous year question papers
2026
2025
2024
2023
2022
2021
2020
2019
2018
2017
2016
2012
2011
2010
2009
2008
2007
2006
2005
2004
2003
2002



