1
JEE Main 2021 (Online) 27th July Morning Shift
Numerical
+4
-1

An organic compound is subjected to chlorination to get compound A using 5.0 g of chlorine. When 0.5 g of compound A is reacted with AgNO3 [Carius Method], the percentage of chlorine in compound A is ____________ when it forms 0.3849 g of AgCl. (Round off to the Nearest Integer)
(Atomic masses of Ag and Cl are 107.87 and 35.5 respectively)
(Atomic masses of Ag and Cl are 107.87 and 35.5 respectively)
Your input ____
2
JEE Main 2021 (Online) 25th July Evening Shift
Numerical
+4
-1

0.8 g of an organic compound was analyzed by Kjeldahl's method for the estimation of nitrogen. If the percentage of nitrogen in the compound was found to be 42%, then ______________ mL of 1 M H2SO4 would have been neutralized by the ammonia evolved during the analysis.
Your input ____
3
JEE Main 2021 (Online) 22th July Evening Shift
Numerical
+4
-1

Methylation of 10 g of benzene gave 9.2 g of toluene. Calculate the percentage yield of toluene __________. (Nearest integer)
Your input ____
4
JEE Main 2021 (Online) 17th March Morning Shift
Numerical
+4
-1

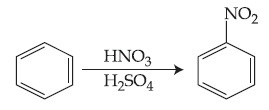
In the above reaction, 3.9 g of benzene on nitration gives 4.92 g of nitrobenzene. The percentage yield of nitrobenzene in the above reaction is ________%. (Round off to the Nearest Integer).
(Given atomic mass : C : 12.0 u, H : 1.0 u, O : 16.0 u, N : 14.0 u)
Your input ____
Questions Asked from Practical Organic Chemistry (Numerical)
Number in Brackets after Paper Indicates No. of Questions
JEE Main 2025 (Online) 7th April Evening Shift (1)
JEE Main 2025 (Online) 4th April Morning Shift (1)
JEE Main 2025 (Online) 2nd April Morning Shift (1)
JEE Main 2025 (Online) 29th January Evening Shift (1)
JEE Main 2025 (Online) 24th January Evening Shift (1)
JEE Main 2024 (Online) 9th April Evening Shift (1)
JEE Main 2024 (Online) 1st February Evening Shift (1)
JEE Main 2024 (Online) 30th January Morning Shift (1)
JEE Main 2023 (Online) 13th April Evening Shift (1)
JEE Main 2023 (Online) 13th April Morning Shift (1)
JEE Main 2022 (Online) 29th July Morning Shift (1)
JEE Main 2022 (Online) 28th July Evening Shift (1)
JEE Main 2022 (Online) 27th July Evening Shift (1)
JEE Main 2022 (Online) 25th July Morning Shift (1)
JEE Main 2022 (Online) 30th June Morning Shift (1)
JEE Main 2022 (Online) 29th June Morning Shift (1)
JEE Main 2022 (Online) 28th June Morning Shift (2)
JEE Main 2022 (Online) 27th June Evening Shift (1)
JEE Main 2022 (Online) 24th June Evening Shift (1)
JEE Main 2021 (Online) 31st August Evening Shift (1)
JEE Main 2021 (Online) 27th August Morning Shift (2)
JEE Main 2021 (Online) 26th August Evening Shift (1)
JEE Main 2021 (Online) 27th July Morning Shift (1)
JEE Main 2021 (Online) 25th July Evening Shift (1)
JEE Main 2021 (Online) 22th July Evening Shift (1)
JEE Main 2021 (Online) 17th March Morning Shift (1)
JEE Main 2021 (Online) 16th March Evening Shift (1)
JEE Main 2021 (Online) 25th February Evening Shift (1)
JEE Main 2020 (Online) 6th September Morning Slot (1)
JEE Main Subjects
Physics
Mechanics
Units & Measurements Vector Algebra Motion in a Straight Line Motion in a Plane Circular Motion Laws of Motion Work Power & Energy Center of Mass and Collision Rotational Motion Properties of Matter Heat and Thermodynamics Simple Harmonic Motion Waves Gravitation
Electricity
Electrostatics Current Electricity Capacitor Magnetic Effect of Current Magnetic Properties of Matter Electromagnetic Induction Alternating Current Electromagnetic Waves
Optics
Modern Physics
Chemistry
Physical Chemistry
Some Basic Concepts of Chemistry Structure of Atom Redox Reactions Gaseous State Chemical Equilibrium Ionic Equilibrium Solutions Thermodynamics Electrochemistry Chemical Kinetics and Nuclear Chemistry Solid State Surface Chemistry
Inorganic Chemistry
Periodic Table & Periodicity Chemical Bonding & Molecular Structure Isolation of Elements Hydrogen s-Block Elements p-Block Elements d and f Block Elements Coordination Compounds Salt Analysis Environmental Chemistry
Organic Chemistry
Mathematics
Algebra
Sets and Relations Logarithm Quadratic Equation and Inequalities Sequences and Series Mathematical Induction Binomial Theorem Matrices and Determinants Permutations and Combinations Probability Vector Algebra 3D Geometry Complex Numbers Statistics Mathematical Reasoning
Trigonometry
Trigonometric Ratio and Identites Trigonometric Equations Inverse Trigonometric Functions Properties of Triangle Height and Distance
Coordinate Geometry
Calculus