1
JEE Advanced 2017 Paper 1 Offline
Numerical
+3
-0

The sum of the number of lone pairs of electrons on each central atom in the following species is
$${[TeB{r_6}]^{2 - }},{\left[ {Br{F_2}} \right]^ + },SNF_3,$$ and $${\left[ {Xe{F_3}} \right]^ - }$$
(Atomic numbers: $$N = 7,F = 9,$$ $$S = 16,Br = 35,$$ $$Te = 52,Xe = 54$$)
$${[TeB{r_6}]^{2 - }},{\left[ {Br{F_2}} \right]^ + },SNF_3,$$ and $${\left[ {Xe{F_3}} \right]^ - }$$
(Atomic numbers: $$N = 7,F = 9,$$ $$S = 16,Br = 35,$$ $$Te = 52,Xe = 54$$)
Your input ____
2
JEE Advanced 2017 Paper 1 Offline
Numerical
+3
-0

Among $${H_2},H{e_2}^ + ,L{i_2},$$ $$B{e_2},{B_2},{C_2},{N_2},O_2^ - $$ and $${F_2},$$ the number of diamagnetic species is
(Atomic numbers : $$H = 1,He = 2,$$ $$Li = 3,Be = 4,$$ $$B = 5,C = 6,$$ $$N = 7,$$ $$O = 8,F = 9$$)
(Atomic numbers : $$H = 1,He = 2,$$ $$Li = 3,Be = 4,$$ $$B = 5,C = 6,$$ $$N = 7,$$ $$O = 8,F = 9$$)
Your input ____
3
JEE Advanced 2017 Paper 1 Offline
Numerical
+3
-0

Among the following, the number of aromatic compound(s) is


Your input ____
4
JEE Advanced 2017 Paper 1 Offline
MCQ (More than One Correct Answer)
+4
-1

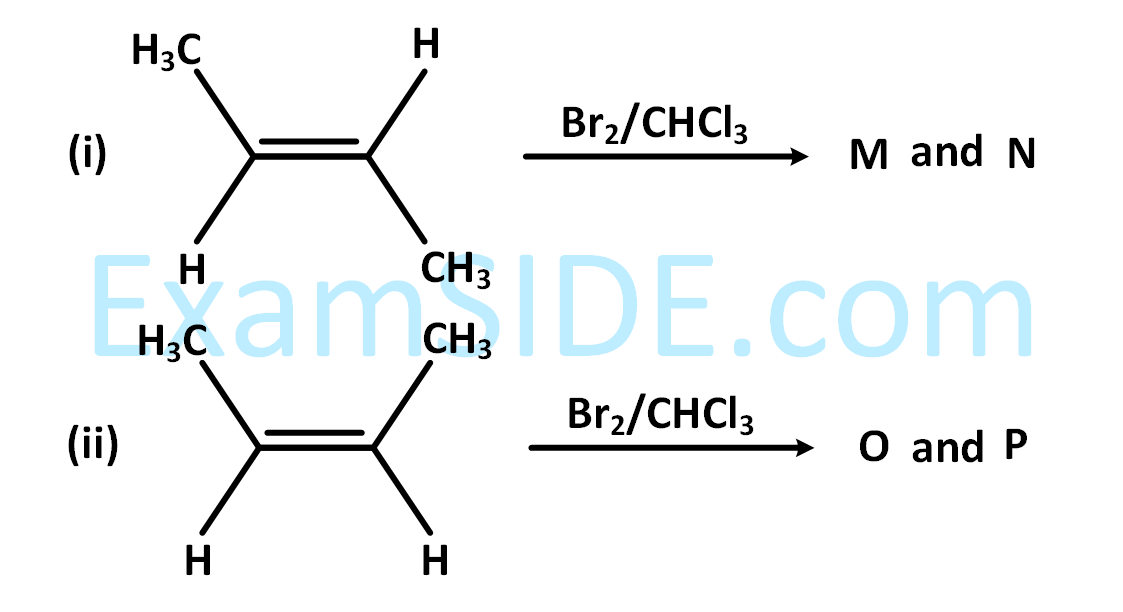
The correct statements(s) for the following addition reactions is (are)

Paper Analysis
Total Questions
Chemistry 18
Mathematics 18
Physics 18
More Papers of JEE Advanced
JEE Advanced 2026 Paper 2 Online JEE Advanced 2026 Paper 1 Online JEE Advanced 2025 Paper 2 Online JEE Advanced 2025 Paper 1 Online JEE Advanced 2024 Paper 2 Online JEE Advanced 2024 Paper 1 Online JEE Advanced 2023 Paper 2 Online JEE Advanced 2023 Paper 1 Online JEE Advanced 2022 Paper 2 Online JEE Advanced 2022 Paper 1 Online JEE Advanced 2021 Paper 2 Online JEE Advanced 2021 Paper 1 Online JEE Advanced 2020 Paper 2 Offline JEE Advanced 2020 Paper 1 Offline JEE Advanced 2019 Paper 2 Offline JEE Advanced 2019 Paper 1 Offline JEE Advanced 2018 Paper 2 Offline JEE Advanced 2018 Paper 1 Offline JEE Advanced 2017 Paper 2 Offline JEE Advanced 2017 Paper 1 Offline JEE Advanced 2016 Paper 2 Offline JEE Advanced 2016 Paper 1 Offline JEE Advanced 2015 Paper 2 Offline JEE Advanced 2015 Paper 1 Offline JEE Advanced 2014 Paper 2 Offline JEE Advanced 2014 Paper 1 Offline JEE Advanced 2013 Paper 2 Offline JEE Advanced 2013 Paper 1 Offline IIT-JEE 2012 Paper 2 Offline IIT-JEE 2012 Paper 1 Offline IIT-JEE 2011 Paper 2 Offline IIT-JEE 2011 Paper 1 Offline IIT-JEE 2010 Paper 2 Offline IIT-JEE 2010 Paper 1 Offline IIT-JEE 2009 Paper 2 Offline IIT-JEE 2009 Paper 1 Offline IIT-JEE 2008 Paper 2 Offline IIT-JEE 2008 Paper 1 Offline IIT-JEE 2007 Paper 2 Offline IIT-JEE 2007 Paper 1 Offline IIT-JEE 2006 IIT-JEE 2005 Screening IIT-JEE 2005 IIT-JEE 2005 Mains IIT-JEE 2004 Screening IIT-JEE 2004 IIT-JEE 2003 Screening IIT-JEE 2003 IIT-JEE 2002 Screening IIT-JEE 2002 IIT-JEE 2001 Screening IIT-JEE 2001 IIT-JEE 2000 IIT-JEE 2000 Screening IIT-JEE 1999 Screening IIT-JEE 1999 IIT-JEE 1998 Screening IIT-JEE 1998 IIT-JEE 1997 IIT-JEE 1996 IIT-JEE 1995 Screening IIT-JEE 1995 IIT-JEE 1994 IIT-JEE 1993 IIT-JEE 1992 IIT-JEE 1991 IIT-JEE 1990 IIT-JEE 1989 IIT-JEE 1988 IIT-JEE 1987 IIT-JEE 1986 IIT-JEE 1985 IIT-JEE 1984 IIT-JEE 1983 IIT-JEE 1982 IIT-JEE 1981 IIT-JEE 1980 IIT-JEE 1979 IIT-JEE 1978
JEE Advanced Papers
All year-wise previous year question papers
2006
1997
1996
1994
1993
1992
1991
1990
1989
1988
1987
1986
1985
1984
1983
1982
1981
1980
1979
1978