1
MHT CET 2025 21st April Morning Shift
MCQ (Single Correct Answer)
+1
-0
What is IUPAC name of the following compound?
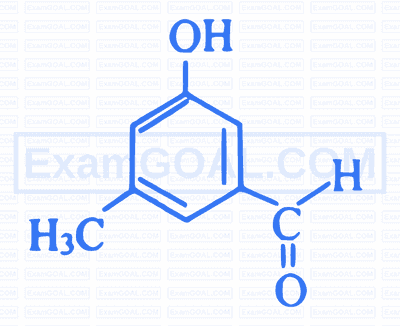
2
MHT CET 2025 21st April Morning Shift
MCQ (Single Correct Answer)
+1
-0
Which of the following compounds has highest boiling point?
3
MHT CET 2025 21st April Morning Shift
MCQ (Single Correct Answer)
+1
-0
Calculate the temperature of 0.05 M sucrose solution in Kelvin if the osmotic pressure of the solution is 1.5 atm .
$$ \left[\mathrm{R}=0.0821 \mathrm{dm}^3 \mathrm{~atm} \mathrm{~K}^{-1} \mathrm{~mol}^{-1}\right] $$
4
MHT CET 2025 21st April Morning Shift
MCQ (Single Correct Answer)
+1
-0
If $\mathrm{E}^*\left(\mathrm{Mg}^{+2}{ }_{(\mathrm{aq})} \mid \mathrm{Mg}_{(\mathrm{s})}\right)=-2.37 \mathrm{~V}$.
What is potential for $\mathrm{Mg}_{(\mathrm{s})} \rightarrow \mathrm{Mg}^{+2}(0.1 \mathrm{M})+2 \mathrm{e^-}$ at 298 K ?
Paper Analysis
Total Questions
Chemistry 50
Mathematics 50
Physics 50
More Papers of MHT CET
MHT CET 2025 5th May Evening Shift MHT CET 2025 26th April Evening Shift MHT CET 2025 26th April Morning Shift MHT CET 2025 25th April Evening Shift MHT CET 2025 25th April Morning Shift MHT CET 2025 23rd April Evening Shift MHT CET 2025 23rd April Morning Shift MHT CET 2025 22nd April Evening Shift MHT CET 2025 22nd April Morning Shift MHT CET 2025 21st April Evening Shift MHT CET 2025 21st April Morning Shift MHT CET 2025 20th April Evening Shift MHT CET 2025 20th April Morning Shift MHT CET 2025 19th April Evening Shift MHT CET 2025 19th April Morning Shift MHT CET 2024 16th May Evening Shift MHT CET 2024 16th May Morning Shift MHT CET 2024 15th May Evening Shift MHT CET 2024 15th May Morning Shift MHT CET 2024 11th May Evening Shift MHT CET 2024 11th May Morning Shift MHT CET 2024 10th May Evening Shift MHT CET 2024 10th May Morning Shift MHT CET 2024 9th May Evening Shift MHT CET 2024 9th May Morning Shift MHT CET 2024 4th May Evening Shift MHT CET 2024 4th May Morning Shift MHT CET 2024 3rd May Evening Shift MHT CET 2024 3rd May Morning Shift MHT CET 2024 2nd May Evening Shift MHT CET 2024 2nd May Morning Shift MHT CET 2023 14th May Evening Shift MHT CET 2023 14th May Morning Shift MHT CET 2023 13th May Evening Shift MHT CET 2023 13th May Morning Shift MHT CET 2023 12th May Evening Shift MHT CET 2023 12th May Morning Shift MHT CET 2023 11th May Evening Shift MHT CET 2023 11th May Morning Shift MHT CET 2023 10th May Evening Shift MHT CET 2023 10th May Morning Shift MHT CET 2023 9th May Evening Shift MHT CET 2023 9th May Morning Shift MHT CET 2022 11th August Evening Shift MHT CET 2021 24th September Evening Shift MHT CET 2021 24th September Morning Shift MHT CET 2021 23rd September Evening Shift MHT CET 2021 23th September Morning Shift MHT CET 2021 22th September Evening Shift MHT CET 2021 22th September Morning Shift MHT CET 2021 21th September Evening Shift MHT CET 2021 21th September Morning Shift MHT CET 2021 20th September Evening Shift MHT CET 2021 20th September Morning Shift MHT CET 2020 19th October Evening Shift MHT CET 2020 16th October Evening Shift MHT CET 2020 16th October Morning Shift MHT CET 2019 3rd May Morning Shift MHT CET 2019 2nd May Evening Shift MHT CET 2019 2nd May Morning Shift
MHT CET Papers
All year-wise previous year question papers
2025
2024
2023
2021
2020