What it the IUPAC name of following compounds?

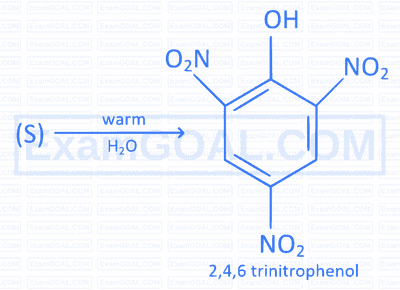
Identify the substrate ' S ' in the following reaction,
Propene on hydroboration followed by oxidation with alkaline peroxide forms.
Calculate the standard enthalpy change of following reaction.
$$ \mathrm{C}_2 \mathrm{H}_{4(\mathrm{~g})}+3 \mathrm{O}_{2(\mathrm{~g})} \longrightarrow 2 \mathrm{CO}_{2(\mathrm{~g})}+2 \mathrm{H}_2 \mathrm{O}_{(l)} $$
If $\Delta_{\mathrm{f}} \mathrm{H}^{\circ}\left(\mathrm{C}_2 \mathrm{H}_4\right)=-52 \mathrm{~kJ} \mathrm{~mol}^{-1}$
$$ \begin{aligned} & \Delta_{\mathrm{f}} \mathrm{H}^{\circ}\left(\mathrm{CO}_2\right)=-390 \mathrm{~kJ} \mathrm{~mol}^{-1} \\ & \Delta_{\mathrm{f}} \mathrm{H}^{\circ}\left(\mathrm{H}_2 \mathrm{O}\right)=-286 \mathrm{~kJ} \mathrm{~mol}^{-1} \end{aligned} $$
MHT CET Papers
All year-wise previous year question papers