Solutions · Chemistry · JEE Advanced
Numerical
At 300 K , an ideal dilute solution of a macromolecule exerts osmotic pressure that is expressed in terms of the height $(h)$ of the solution (density $=1.00 \mathrm{~g} \mathrm{~cm}^{-3}$ ) where $h$ is equal to 2.00 cm . If the concentration of the dilute solution of the macromolecule is $2.00 \mathrm{~g} \mathrm{dm}^{-3}$, the molar mass of the macromolecule is calculated to be $\boldsymbol{X} \times 10^4 \mathrm{~g} \mathrm{~mol}^{-1}$. The value of $\boldsymbol{X}$ is __________.
Use: Universal gas constant $(R)=8.3 \mathrm{~J} \mathrm{~K}^{-1} \mathrm{~mol}^{-1}$ and acceleration due to gravity $(g)=10 \mathrm{~m} \mathrm{~s}^{-2}$
The elevation of boiling point for solution in Vessel-1 is ________ $\%$ of the solution in Vessel-2.
[Use: Molar mass of urea $=60 \mathrm{~g} \mathrm{~mol}^{-1}$; gas constant, $\mathrm{R}=62$ L Torr $\mathrm{K}^{-1} \mathrm{~mol}^{-1}$;
Assume, $\Delta_{\text {mix }} \mathrm{H}=0, \Delta_{\text {mix }} \mathrm{V}=0$ ]
(Given data: Molar mass and the molal freezing point depression constant of benzene are 78 g mol-1 and 5.12 K kg mol-1, respectively).

On addition of equal number of moles of a non-volatile solute $$S$$ in equal amount (in $$kg$$) of these solvents, the elevation of boiling point of solvent $$X$$ is three times that of solvent $$Y$$. Solute $$S$$ is known to undergo dimerization in these solvents. If the degree of dimerization is $$0.7$$ in solvent $$Y$$, the degree of dimerization in solvent $$X$$ is ___________.
(given that the vapor pressure of pure liquid $$A$$ is $$20$$ $$Torr$$ at temperature $$T$$)
MCQ (Single Correct Answer)
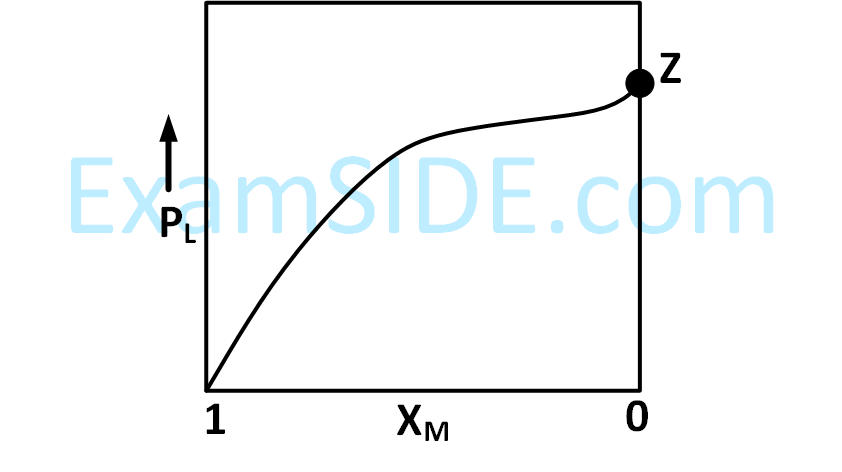
The qualitative sketches I, II and III given below show the variation of surface tension with molar concentration of three different aqueous solutions of KCl, CH3OH and CH3(CH2)11 OSO$$_3^ - $$ Na+ at room temperature. The correct assignment of the sketches is

The Henry's law constant for the solubility of N$$_2$$ gas in water at 298 K is 1.0 $$\times$$ 10$$^5$$ atm. The mole fraction of N$$_2$$ in air is 0.8. The number of moles of N$$_2$$ from air dissolved in 10 moles of water at 298 K and 5 atm pressure is
The freezing point of the solution M is :
The vapour pressure of the solution M is :
Water is added to the solution M such that the fraction of water in the solution becomes 0.9 mole. The boiling point of this solution is:
MCQ (More than One Correct Answer)