Biology
1. Megaspores are produced from the megaspore mother cells after 2. The pineapple which under natural condition is difficult to blossom has been made to produce fruits throughout the year 3. During muscle contraction in humans, the 4. Select the option which shows correct matching of animal with its excretory organ and excretory product. 5. Select the correct statement with respect to disorders of muscles in humans. 6. Select the option which correctly matches the endocrine gland with its hormone and its function. 7. Norepinephrine
(i) is released by sympathetic fibres
(ii) is released by parasympathetic fibers
( 8. Which of the following represents the action of insulin? 9. Animal vectors are required for pollination in 10. In our society women are blamed for producing female children. Choose the correct answer for the sex-determination in hu 11. Which one of the following statements is correct? 12. Albuminous seeds store their reserve food mainly in 13. The foetal ejection reflex in humans triggers the release of 14. Which of the following statements is correct? 15. Which one is the incorrect statement with regards
to the importance of pedigree analysis? 16. The stage transferred into the uterus after induced fertilization of ovum in the laboratory is 17. Down's syndrome in humans is due to : 18. One of the following is not a method of contraception. Which one? 19.
The figure gives an important concept in the genetic
implication of DNA . Fill the blanks A, B and C. 20. Genes of interest can be selected from a genomic
library by using : 21. Among flowers of Calotropis, tulip, Sesbania, Asparagus, Colchicum, sweet pea, Petunia, Indigofera, mustard, soybean, t 22. What is common in all the three, Funaria, Dryopteris and Ginkgo? 23. Which one of the following is true for fungi? 24. Which one of the following is wrongly matched? 25. Why is a capsule advantageous to a bacterium? 26. The plant body is thalloid in 27. Which one of the following groups of animals reproduces only by sexual means? 28. The characteristics of Class Reptilia are 29. Which one of the following is one of the paths followed by air or O2 during respiration in the adult male Periplaneta am 30. Sharks and dogfishes differ from skates and rays because 31. Which one of the following animals is correctly matched with its one characteristic and the taxon? 32. Which of the following is not a property of the
genetic code ? 33. Inflorescence is racemose in 34. How many plants among China rose, Ocimum, sunflower, mustard, Alstonia, guava, Calotropis and Nerium (oleander) have opp 35. In a cymose inflorescence the main axis 36. During the metaphase stage of mitosis, spindle fibres attach to chromosomes at 37. Uridine, present only in RNA is a 38. Which of the following statements about enzymes is wrong? 39. The figure shows a hypothetical tetrapeptide portion of a protein with parts labelled A - D. Which one of the following 40. During meiosis I, the chromosomes start pairing at 41. Identify the tissue shown in the diagram and match
with its characteristics and its location :-
42. Which of the following has maximum genetic
diversity in India? 43. When man eats fish which feeds on zooplankton
which have eaten small plants, the producer in the
chain is : 44. Which two distinct microbial processes are
responsible for the release of fixed nitrogen as
dinitrogen gas (N2) to the a 45. Which one of the following is a primary consumer
in maize field ecosystem ? 46. Select the correct option with respect to
Cockroaches :- 47. Bundle sheath cells : 48. Which of the following statements is not true for
stomatal apparatus ? 49. Meristematic tissue responsible for increase in girth
of tree trunk is :
50. Which organization publishes the 'Red Data
Book' ? 51. A stage of mitosis is shown in the diagram.
Which stage is it and what are its characteristics?
52. The term 'glycocalyx' is used for : 53. Which of the following best illustrates "FEEDBACK"
in development ? 54. Which of the following types of plastids does not
contain stored food material ? 55. Select the alternative giving correct identification
and function of the organelle 'A' in the diagram
56. The figure shows a section of human ovary.
Select the option which gives the correct
identification of either A or B wit 57. A sagittal section of human brain is shown here.
Identify at least two labels from A-D.
58. The figure shows blood circulation in humans with
labels A to D. Select the option which gives correct
identification of 59. The figure shows a human blood cell. Identify
it and give its characteristics.
60. The figure shows an axon terminal and synapse.
Select the option giving correct identification
of labels A-D.
61. Microbe used for biocontrol of pest butterfly
caterpillars is : 62. In an inducible operon, the genes are : 63. Satellite RNA are present in some : 64. One of the most frequently used techniques in DNA
fingerprinting is :- 65. Random undirectional change in allele frequencies
that occur by chance in all populations and especially
in small popula 66. Genetic variation in a population arises due to : 67. Dinosaurs dominated the World in which of the
following geological era ? 68. The finch species of Galapagos Islands are grouped
according to their food sources. Which of the
following is not a finc 69. Which one of the following statements is correct
regarding Sexually Transmitted Diseases (STD) ? 70. Which of the following statements is not true about
somatic embryogenesis ? 71. Which one of the following is a hallucinogenic drug? 72. Syngamy can occur outside the body of the organism in 73. Identify the site where Wuchereria bancrofti is
normally found on human body : 74. Tissue culture technique can produce infinite
number of new plants from a small parental tissue.
The economic importance 75. RNA interference involves :- 76. The age pyramid with broad base indicates : 77. During the process of isolation of DNA, chilled
ethanol is added to : 78. Which one of the following is not a parasitic
adaptation ? 79. Which one of the following vectors is used to replace
the defective gene in gene therapy ? 80. Benthic organisms are affected most by : 81. The largest tiger reserve in India is :- 82. The common characteristics between tomato and potato will be maximum at the level of their
Chemistry
1. According to law of photochemical equivalence the energy absorbed (in ergs/mole) is given as (h = 6.62 $$ \times $$ 10$$ 2. On hydrolysis of a ''compound'', two compounds are obtained. One of which one treatment with sodium nitrite and hydrochl 3. Some reactions of amines are given. Which one is not correct? 4. In DNA, the linkages between different nitrogenous bases are 5. Which condition is not satisfied by an ideal solution? 6. For a reaction between A and B the order with respect to A is 2 and the other with respect to B is 3. The concentrations 7. Consider the half-cell reduction reaction
Mn2+ + 2e$$-$$ $$ \to $$ Mn, Eo = $$-$$1.18 V
Mn2+ $$ \to $$ Mn3+ + e$$-$$, 8. A reaction is 50% complete in 2 hours and 75% complete in 4 hours. The order of reaction is 9. How many gram of cobalt metal will be deposited when a solution of cobalt (II) chloride is electrolyzed with a current o 10. Which statement is wrong? 11. Identify the incorrect statement, regarding the molecule XeO4 : 12. Number of isomeric alcohols of molecular formula C6H14O which give positive iodoform test is 13. Sc (Z = 21) is a transition element but Zn (Z = 30) is not because
14. In a particular isomer of [Co(NH3)4Cl2]o, the Cl$$-$$Co$$-$$Cl angle is 90o, this isomer is known as 15. Which is diamagnetic? 16. The correct IUPAC name for [CrF2(en)2]Cl is 17. Crystal field splitting energy for high spin d4 octahedral complex is 18. Which among the following is a paramagnetic complex? 19. The anion of acetylacetone (acac) forms Co (acac)3 chelate with Co3+. The rings of the chelate are 20. Accumulation of lactic acid (HC3H5O3), a monobasic acid in tissues leads to pain and a feeling of fatigue. In a 0.10 M a 21. The outer electronic configuration of Gd (At. No. 64) is 22. Which one of the following arrangements represents the correct order of least negative to most negative electron gain en 23. In an experiment it showed that 10 mL of 0.05 M solution of chloride required 10 mL of 0.1 M solution of AgNO3, which of 24. In which of the following ionization processes the bond energy increases and the magnetic behaviour changes from paramag 25. In which of the following pair both the species have sp3 hybridization ? 26. The pair of species that has the same bond order in the following is 27. The outer orbitals of C in ethane molecule can be considered to be hybridized to give three equivalent sp2 orbitals. The 28. When 5 litres of a gas mixture of methane and propane is perfectly combusted at 0oC and 1 atmosphere, 16 litres of oxyge 29. Three thermochemical equations are given below
(i) C(graphite) + O2(g) $$ \to $$ CO2(g); $$\De 30. The dissociation constant of weak acid is 1 $$ \times $$ 10$$-$$4. In order to prepare a buffer solution with a pH = 5, 31. Arrange the following in increasing order of stability
32. Homolytic fission of the following alkanes forms free radicals CH3 $$-$$ CH3, CH3 $$-$$ CH2 $$-$$ CH3, (CH3)2CH $$-$$ CH 33. The values of Ksp of CaCO3 and CaC2O4 are 4.7 $$ \times $$ 10$$-$$9 and 1.3 $$ \times $$ $$-$$9 respectively at 25oC. I 34. What is the hybridisation state of benzyl carbonium ion?
35. At 100oC the Kw of water is 55 times its value at 25oC. What will be the pH of neutral solution? (log 55 = 1.74) 36. Nitrogen detection in an organic compound is carried out by Lassaigne's test. The blue colour formed corresponds to whic 37. Which of the following chemical system is non aromatic ? 38. In the following reaction :
Product 'P' will not give 39. Given :
I and II are
Physics
1. The de-broglie wavelength of neutrons in thermal equilibrium at temperature T is 2. Two rods are joined end to end, as shown. Both have a cross-sectional area of 0.01 cm2. Each is 1 meter long. One rod is 3. A charge q is placed at the centre of the line joining two equal charges Q. The system of the three charges will be in e 4. A 12 cm wire is given a shape of a right angled triangle ABC having sides 3 cm, 4 cm and 5 cm as shown in the figure. Th 5. Ten identical cells connected in series are needed to heat a wire of length one meter and radius 'r' by 10oC in time 't' 6. A bar magnet of magnetic moment M is placed at right angles to a magnetic induction B. If a force F is experienced by ea 7. A circular coil ABCD carrying a current 'i' is placed in a uniform magnetic field. If the magnetic force on the segment 8. A long straight wire carries a certain current and produces a magnetic field 2 $$ \times $$ 10$$-$$4 Wb m$$-$$2 at a per 9. An electromagnetic wave of frequency $$\upsilon = 3.0$$ MHz passes from vaccum into a dielectric medium with relative p 10. The primary of a transformer when connected to a dc battery of 10 Volt draws a current of 1 mA. The number of turns of t 11. A current of 2.5 A flows through a coil of inductance 5 H. The magnetic flux linked with the coil is 12. A source of light is placed at a distance of 50 cm from a photo cell and the stopping potential is found to be V0. If th 13. An electric dipole of dipole moment p is aligned parallel to a uniform electric field E. The energy required to rotate t 14. In Young's double slit experiment the distance between the slits and the screen is doubled. The separation between the s 15. Two plane mirrors are inclined at 70o. A ray incident on one mirror at angle, $$\theta $$ after reflection falls on seco 16. A parallel beam of light of wavelength $$\lambda $$ is incident normally on a narrow slit. A diffraction pattern formed 17. The reddish appearance of the sun at sunrise and sunset is due to 18. How does the Binding Energy per nucleon vary with the increase in the number of nucleons ? 19. An electron in hydrogen atom makes a transition n1 $$ \to $$ n2 where n1 and n2 are principal quantum numbers of the two 20. One way in which the operation of a n-p-n transistor differs from that of a p-n-p 21. The output from a NAND gate is divided into two in parallel and fed to another NAND gate. The resulting gate is a
22. In an unbiased p-n junction, holes diffuse from the p-region to n-region because of 23. If the ratio of diameters, lengths and Young's modulus of steel and copper wires shown in the figure are p, q and s resp 24. The pair of quantities having same dimensions is 25. Vectors $$\overrightarrow A ,\overrightarrow B $$ and $$\overrightarrow C $$ are such that $$\overrightarrow A .\overrig 26. One coolie takes 1 minute to raise a suitcase through a height of 2 m but the second coolie takes 30 s to raise the same 27. A car is moving in a circular horizontal track of radius 10 m with a constant speed of 10 m/s. A bob is suspended from t 28. A person holding a rifle (mass of person and rifle together is 100 kg) stands on a smooth surface and fires 10 shots hor 29. A particle with total energy E is moving in a potential energy region U(x). Motion of the particle is restricted to the 30. The ratio of radii of gyration of a circular ring and a circular disc, of the same mass and radius, about an axis passin 31. Two discs are rotating about their axes, normal to the discs and passing through the centres of the discs. Disc D1 has 2 32. A particle of mass 'm' is kept at rest at a height 3R from the surface of earth, where 'R' is radius of earth and 'M' is 33. The radius of a planet is twice the radius of earth. Both have almost equal average mass densities. VP and VE are escape 34. Two metal rods 1 and 2 of same lengths have same temperature difference between their ends. Their thermal conductivities 35. A fluid is in streamline flow across a horizontal pipe of variable area of cross section. For this which of the followin 36. The density of water at 20oC is 998 kg/m3 and at 40oC is 992 kg/m3. The coefficient of volume expansion of water is 37. Two Carnot engines A and B are operated in series. The engine A receives heat from the source at temperature T1 and reje 38. Which of the following relations does not give the equation of an adiabatic process, where terms have their usual meanin 39. A system is taken from state a to state c by two paths adc and abc as shown in the figure. The internal energy at a is $ 40. In a vessel, the gas is at pressure P. If the mass of all the molecules is halved and their speed is doubled, then the r 41. The length of the wire between two ends of a sonometer is 100 cm. What should be the positions of two bridges below the 42. A particle of mass m oscillates along x-axis according to equation x = asin$$\omega $$t. The nature of the graph between 43. The displacement 'x' (in meter) of a particle of mass 'm' (in kg) moving in one dimension under the action of a force, i
1
NEET 2013 (Karnataka)
MCQ (Single Correct Answer)
+4
-1

The de-broglie wavelength of neutrons in thermal equilibrium at temperature T is
A
$${{3.08} \over {\sqrt T }}\mathop A\limits^ \circ $$
B
$${{0.308} \over {\sqrt T }}\mathop A\limits^ \circ $$
C
$${{0.0308} \over {\sqrt T }}\mathop A\limits^ \circ $$
D
$${{30.8} \over {\sqrt T }}\mathop A\limits^ \circ $$
2
NEET 2013 (Karnataka)
MCQ (Single Correct Answer)
+4
-1

Two rods are joined end to end, as shown. Both have a cross-sectional area of 0.01 cm2. Each is 1 meter long. One rod is of copper with a resistivity of 1.7 $$ \times $$ 10$$-$$6 ohm-centimeter, the other is of iron with a resistivity of 10$$-$$5 ohm-centimeter.
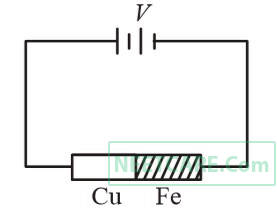
How much voltage is required to produce a current of 1 ampere in the rods?

How much voltage is required to produce a current of 1 ampere in the rods?
A
0.00145 V
B
0.0145 V
C
1.7 $$ \times $$ 10$$-$$6 V
D
0.117 V
3
NEET 2013 (Karnataka)
MCQ (Single Correct Answer)
+4
-1

A charge q is placed at the centre of the line joining two equal charges Q. The system of the three charges will be in equilibrium if q is equal to
A
$$-$$Q/4
B
Q/4
C
$$-$$Q/2
D
Q/2
4
NEET 2013 (Karnataka)
MCQ (Single Correct Answer)
+4
-1

A 12 cm wire is given a shape of a right angled triangle ABC having sides 3 cm, 4 cm and 5 cm as shown in the figure. The resistance between two ends (AB, BC, CA) of the respective sides are measuread one by one ratio
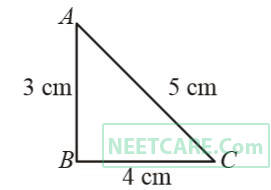

A
9 : 16 : 25
B
27 : 32 : 35
C
21 : 24 : 25
D
3 : 4 : 5
Paper Analysis
Total Questions
Biology 82
Chemistry 39
Physics 43
More Papers of NEET
NEET 2026 NEET 2025 NEET 2024 (Re-Examination) NEET 2024 NEET 2023 Manipur NEET 2023 NEET 2022 Phase 2 NEET 2022 Phase 1 NEET 2021 NEET 2020 Phase 1 NEET 2019 NEET 2018 NEET 2017 NEET 2016 Phase 2 NEET 2016 Phase 1 AIPMT 2015 AIPMT 2015 Cancelled Paper AIPMT 2014 NEET 2013 (Karnataka) NEET 2013 AIPMT 2012 Mains AIPMT 2012 Prelims AIPMT 2011 Mains AIPMT 2011 Prelims AIPMT 2010 Mains AIPMT 2010 Prelims AIPMT 2009 AIPMT 2008 AIPMT 2007 AIPMT 2006 AIPMT 2005 AIPMT 2004 AIPMT 2003 AIPMT 2002 AIPMT 2001 AIPMT 2000
NEET Papers
All year-wise previous year question papers
2014
2009
2008
2007
2006
2005
2004
2003
2002
2001
2000